Strategies to Reduce Animal Testing in US EPA's HPV Programme
An Animal Rights Article from All-Creatures.org
FROM
Chad B. Sandusky and Megha Even,
Physicians Committee, and Kristie Stoick
and Jessica Sandler (PETA)
January 2007
Summary
The High Production Volume (HPV) programme was launched by the US Environmental Protection Agency (EPA) in 1998. To reduce animals killed, the animal welfare community negotiated basic principles set forth in a letter from EPA to HPV participants, and reiterated in the Federal Register (2000). After five years reviewing more than 370 test plans, the outcome of this effort is disappointing. However, successful strategies were developed by these reviewers and in collaboration with conscientious companies to reduce testing and still meet the Screening Information Data Set (SIDS) requirements. These strategies are explored as they might apply to future testing programmes.
Keywords: high production volume (HPV), three Rs, chemical testing, toxicity testing, animal testing
Introduction and historical overview
The EPAs HPV programme has been in progress for five years, and was envisioned to involve commitments for 2,200 chemicals by 400 companies. Submitters were to assess existing hazard data and data gaps, and propose a plan to fill these perceived gaps. Originally designated as a voluntary programme, since its inception hundreds of test plans have been submitted, many of which propose animal tests to complete the SIDS base set of Tier I data requirements. The HPV programme was modeled after a similar programme administered by the OECD, i.e., the Task Force on Existing Chemicals. In the U.S. programme, the SIDS is considered a minimum for hazard evaluation and hazard is stressed over considerations of potential exposure. While each of the 3Rs (replacement, reduction, refinement) is available to HPV participants, they have frequently been ignored and/or followed to varying degrees. This current paper provides an update to a previous presentation at the Fourth World Congress on Alternatives and Animal use in the Life Sciences, in which the US HPV programme was critiqued shortly after its implementation (Nicholson et. al., 2004).
Screening information data set (SIDS)
The Tier I SIDS data requirements which use animals are provided in table 1, along with the corresponding OECD Test Guideline number and the number of animals used for each test.
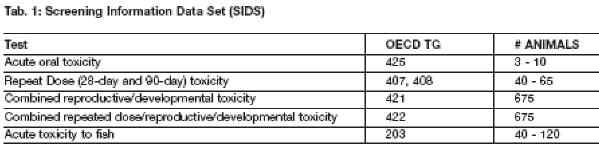
The numbers of animals used per test can vary depending on exact study design, but the total number killed for a complete data set ranges from 758-870 mice, rats, and fish. Although an exact number is not possible to calculate, we have estimated that since the inception of the HPV programme, upwards of 150,000 animals have already been killed (through April 2005).
Animal welfare guidance and principles
When animal protection organisations became aware of the proposed programme, they maintained that the programme objectives, primarily the protection of human health, would not be met and that the cost in animal lives would be exorbitant. Through the White House, they also negotiated with stakeholders (EPA/Environmental Defense/American Chemistry Council) was the October 1999 agreement letter, sent by EPA to programme participants (Wayland, 1999). These principles were subsequently published in the Federal Register (2000). The main tenets of these principles encouraged the following:
- The use of in vitro genetic toxicity testing rather than in vivo (unless impossible).
- Maximising the use of existing data.
- The use of weight-of-evidence and avoid checklist toxicology. The use of Structure Activity Relationships (SAR) to form chemical categories.
- No terrestrial resting (e.g., birds, etc).
- No new dermal testing (generally).
- No sub-chronic or reproductive toxicity testing on closed system intermediates.
- Special considerations for chemicals which have been previously determined to be GRAS (Generally Recognised As Safe).
- The use of validated non-animal tests as they become available and the delay of certain testing until some non-animal methods were in place.
The goal of these animal welfare principles was to minimise animal use, while still meeting the stated hazard identification goals of the programme. Our aim was to assist companies in avoiding check-the-box toxicology to fulfill the basic SIDS data set. If the recommended generalised principles of the October 1999 letter were indeed followed, the result would be a reduction in the numbers of animals killed under the HPV programme.
Each of the 3R principles (replacement, reduction and refinement) was available to HPV participants. Reductions in the numbers of animals could be accomplished, for example, by using categories of chemicals to maximize existing data or by using established OECD combined protocols such as the OECD TG 422, a combined repeat-dose, reproductive, and developmental toxicity screen, instead of three separate tests to fulfill the endpoints.
Refinements to tests involving animals included the use of OECD TG 425 and cytotoxicity tests instead of the traditional LD50. Finally, replacing animals completely was possible in some cases, such as the use of in vitro genetic toxicity tests rather than in vivo. The principle of thoughtful toxicology, outlined in the October 1999 letter, provided an overarching opportunity for companies to carefully analyse existing data and decide whether additional animal tests would provide information that would be useful or relevant and to avoid such testing where it would not.
What went wrong with implementation of the HPV programme?
Once initiated, it became clear that the sponsors of HPV test plans often failed to follow even the minimal guidance offered above. The guidelines were not enforceable, and there is still no mechanism in 2005 to ensure that animal welfare guidelines are followed. In many cases, companies duplicated testing unnecessarily by conducting animal tests that had already been conducted but werent conducted following Good Laboratory Practices (GLP), or by failing to coordinate efforts with other companies sponsoring similar chemicals. In other instances, companies did not use existing published data, individually or in conjunction with other data (in a weight-of-evidence approach), to avoid new animal testing.
Often times, sponsors would fail to show relevance, such as proposing acute fish toxicity tests on water-insoluble chemicals. In many cases, when it was clear that a test was not needed for HPV, the study was proposed in anticipation of future data requirements, primarily Registration, Evaluation and Authorisation of Chemicals (REACH). Some companies refused to use combined protocols, sometimes doubling the number of animals killed under their test plans. Even in obvious replacement opportunities, such as the use of in vitro genotoxicity tests, there was an inconsistent application of the principle. In its responses to test plan proposals, EPA itself frequently failed to follow or encourage sponsors to follow basic animal welfare guidance.
HPV since 2000
Scientists at PCRM and PETA have reviewed approximately 376 test plans through August 2005, representing both individual chemicals and small to large groups of chemicals. According to our figures, a full 50% of the test plans proposed from 2000 through 2002 called for animal testing, another 50% of the test plans submitted in 2003, 45% of the test plans in 2004, and 33% of the test plans submitted through May 2005 proposed new animal testing.
These test plans account for more than 150,000 animals used to date. Importantly, these figures do not include animal tests requested by EPA above and beyond those proposed in the original test plans. It is noteworthy that after more than five years and 150,000 animals killed, no additional protections have been implemented to protect human health or the environment as a result of the HPV programme. Importantly, hazard data being generated offer little in the way of assessing human risk in that exposure characterisations are discouraged and to some degree specifically excluded from the programme. Thus, there is no context to assess the large amount of hazard information being generated by the sponsors.
Additional strategies to reduce animal use in the HPV programme
In the process of reviewing hundreds of test plans, additional strategies have been developed to supplement those envisioned in the original October 1999 letter. Some of these are extensions of the original recommendations, e.g., common sense toxicology, identification of duplicative testing and/or overlooked data, promoting stronger weight-of evidence approaches, etc. In addition, the wise use of resources has been stressed (a full SIDS data set may cost up to $400,000 USD) as well as encouraging companies to resist regulatory pressures when testing does not make sense. In addition to the existing guidelines, and based on an extensive review of HPV test plans, additional animal welfare principles are described below.
- Rapid Hydrolysis of Parent Chemical. The parent chemical need not be tested in animals if it hydrolyses to well-characterised products in an aqueous environment at low pH. A bench study at stomach pH may be used to determine rate of hydrolysis and hydrolytic products. Existing data on the hydrolysis products may then be used to meet SIDS endpoints without additional testing.
- Acidic/Corrosive/Irritating Materials. These are usually strong acids; they may be completely ionised in aqueous environments and are expected to cause localised, corrosive effects in the GI tract. Results from animal tests will be confounded by the corrosivity of the chemical and mammalian testing would not yield meaningful results. Animal tests using such material are particularly painful.
- Highly Reactive Materials. These chemicals are highly reactive to air and/or water as demonstrated by physical/ chemical data. Mammalian and ecotoxicity testing with these types of chemicals is not feasible.
- Gases. Primary concerns with these chemicals are flammability, explosivity at test levels, and/or insolubility in water. Many are asphyxiants, some are minimally toxic and rapidly excreted, so additional testing may not be feasible or will not yield meaningful results.
- Complex Mixtures. The product is a mixture from different manufacturing processes and/or waste streams. Additional testing with a variable mixture does not provide useful information and existing data on major constituents may be sufficient to fill SIDS endpoints.
- Weight-of-Evidence. Additional testing for reproductive toxicity can be eliminated if histopathology data on reproductive organs from a 90-day subchronic study are available, in combination with a negative developmental study. In some cases, traditional reproduction/ developmental studies are not required if existing data from other studies, such as 2-year cancer bioassays, have evaluated reproductive and developmental parameters. A separate developmental study is not required if data exists from one- or two-generation reproduction studies. This guidance is provided in the Manual for Investigation of HPV Chemicals OECD Secretariat (SIDS Manual, 2004).
These additional strategies have been employed successfully in the US HPV programme, and have resulted in saving thousands of animals.
Implementation and Implications
In order to implement these strategies, much time is spent by PCRM and PETA reviewing each test plan, conducting internet data searches, submitting detailed comments during the public comment period, and finally, contacting individual companies to discuss opportunities to eliminate or at least reduce animal testing. We encourage companies to submit revised test plans, and we offer support to those that have already used creative and well developed strategies that reduce testing (sometimes in the form of letters to the EPA). Continued review and comment will hopefully result in future opportunities to reduce animal testing further, both in the impending REACH programme and the recently announced Extended HPV programme, planned for a January 2006 initiation and running through 2010.
References
Federal Register, Vol. 65, No. 248, December 26, 2000, pp. 81686-98.
Nicholson, A., Sandler, J. and Seidle, T. (2004). An evaluation of the US High Production Volume (HPV) Chemical-testing Programme: A Study in (Ir)Relevance, Redundancy and Retrothinking. ATLA 32, Supplement 1, 335-341, Fourth World Congress.
SIDS Manual..
Wayland, S. (1999). Letter to HPV programme participants, 2pp. Washington, DC, USA: US Environmental Agency.
Return to Animal Rights Articles
Read more at Alternatives to Animal Testing, Experimentation and Dissection